According to the article of David E. Clapham published in PNAS the 02/ 11/ 2021 and disclosed in #OKILAB
Mammalian sperm hyperactivation regulates navigation via physical boundaries and promotes pseudo-chemotaxis
Significance
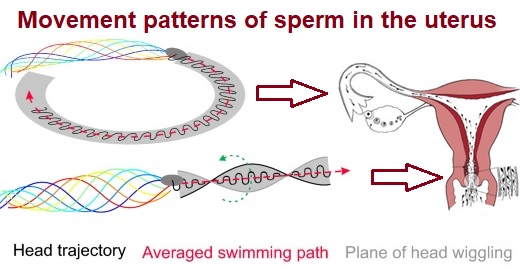
Mammalian sperm migration within the female reproductive tract requires navigational mechanisms, through which sperm respond to biophysical and biochemical cues. Previous studies revealed that biophysical cues for sperm in the female reproductive tract include fluid flow, wall architecture, and temperature gradients. Here, by exploring bovine sperm motion in microfluidic reservoirs and by developing theoretical and computational models, we demonstrate that sperm motility hyperactivation, which is part of a process that prepares sperm in the female tract for fertilization and involves an increase in the asymmetry of sperm flagellar beating, modulates sperm–sidewall interactions and thus navigation via female tract wall architecture. Specifically, hyperactivation reduces the tendency of sperm to remain swimming along walls and promotes a response that resembles chemotaxis.
Abstract
Mammalian sperm migration within the complex and dynamic environment of the female reproductive tract toward the fertilization site requires navigational mechanisms, through which sperm respond to the tract environment and maintain the appropriate swimming behavior. In the oviduct (fallopian tube), sperm undergo a process called “hyperactivation,” which involves switching from a nearly symmetrical, low-amplitude, and flagellar beating pattern to an asymmetrical, high-amplitude beating pattern that is required for fertilization in vivo. Here, exploring bovine sperm motion in high–aspect ratio microfluidic reservoirs as well as theoretical and computational modeling, we demonstrate that sperm hyperactivation, in response to pharmacological agonists, modulates sperm–sidewall interactions and thus navigation via physical boundaries. Prior to hyperactivation, sperm remained swimming along the sidewalls of the reservoirs; however, once hyperactivation caused the intrinsic curvature of sperm to exceed a critical value, swimming along the sidewalls was reduced. We further studied the effect of noise in the intrinsic curvature near the critical value and found that these nonthermal fluctuations yielded an interesting “Run–Stop” motion on the sidewall. Finally, we observed that hyperactivation produced a “pseudo-chemotaxis” behavior, in that sperm stayed longer within microfluidic chambers containing higher concentrations of hyperactivation agonists