According to the article of Wei Wei published in Science the 24 / 05 / 2019 and disclosed in #OKILAB
Germline selection shapes human mitochondrial DNA diversity
Heteroplasmy incidence in mitochondrial DNA
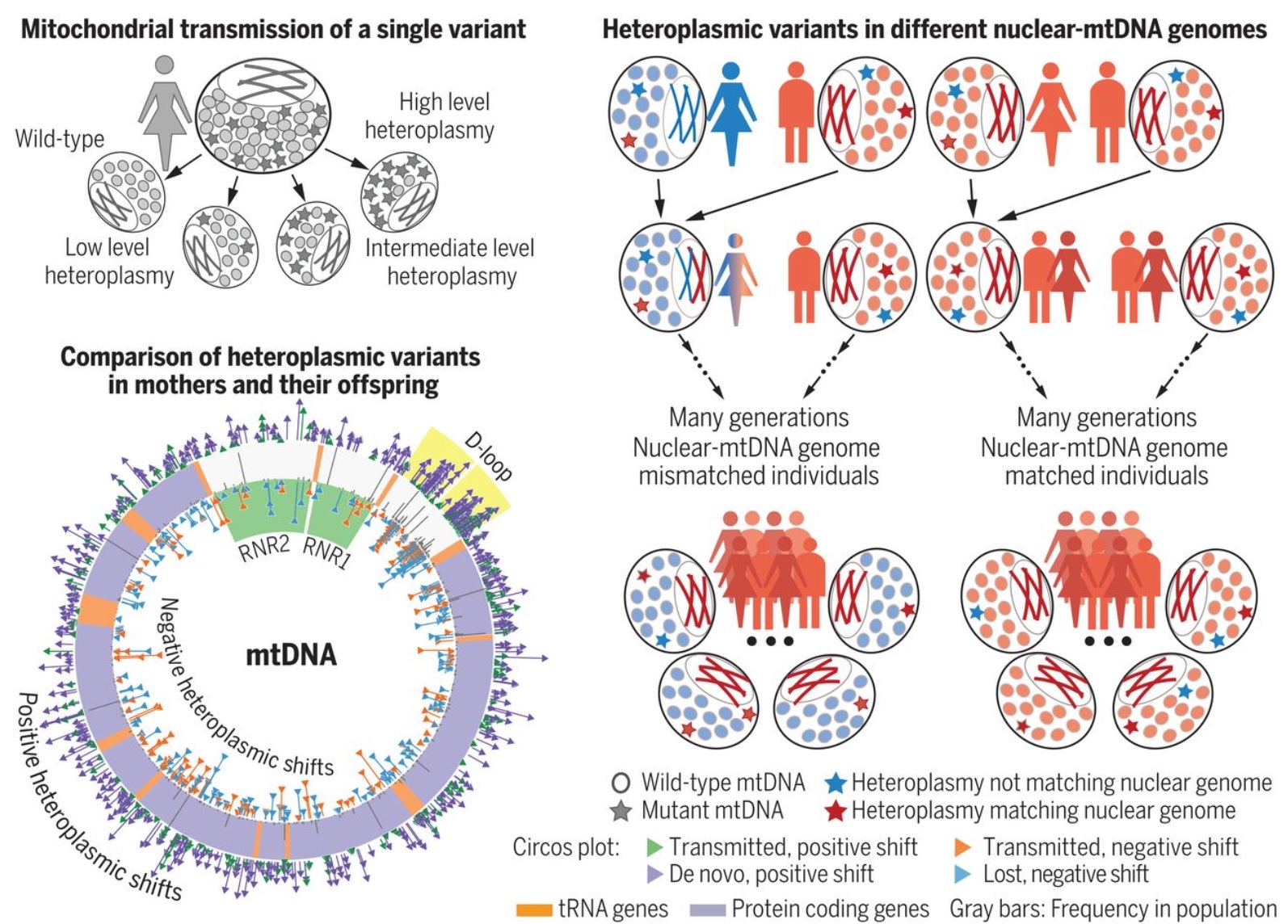
In humans, mitochondrial DNA (mtDNA) is predominantly maternally inherited. mtDNA is under selection to prevent heteroplasmy—the transmission of multiple genetic variants into the next generation. Wei et al. explored human mtDNA sequences to determine mtDNA genome structure, selection, and transmission. Whole-genome sequencing revealed that about 45% of individuals carry heteroplasmic mtDNA sequences at levels greater than 1% of their total mtDNA. Furthermore, studies of more than 1500 mother-offspring pairs indicated that the female line selected which mtDNA variants were passed on to children. This effect was influenced by the mother’s nuclear genetic background. Thus, mtDNA is under selection at specific loci in the human germ line.
Science, this issue p. eaau6520
Structured Abstract
INTRODUCTION
Only 2.4% of the 16.5-kb mitochondrial DNA (mtDNA) genome shows homoplasmic variation at >1% frequency in humans. Migration patterns have contributed to geographic differences in the frequency of common genetic variants, but population genetic evidence indicates that selection shapes the evolving mtDNA phylogeny. The mechanism and timing of this process are not clear.
Unlike the nuclear genome, mtDNA is maternally transmitted and there are many copies in each cell. Initially, a new genetic variant affects only a proportion of the mtDNA (heteroplasmy). During female germ cell development, a reduction in the amount of mtDNA per cell causes a “genetic bottleneck,” which leads to rapid segregation of mtDNA molecules and different levels of heteroplasmy between siblings. Although heteroplasmy is primarily governed by random genetic drift, there is evidence of selection occurring during this process in animals. Yet it has been difficult to demonstrate this convincingly in humans.
RATIONALE
To determine whether there is selection for or against heteroplasmic mtDNA variants during transmission, we studied 12,975 whole-genome sequences, including 1526 mother–offspring pairs of which 45.1% had heteroplasmy affecting >1% of mtDNA molecules. Harnessing both the mtDNA and nuclear genome sequences, we then determined whether the nuclear genetic background influenced mtDNA heteroplasmy, validating our findings in another 40,325 individuals.
RESULTS
Previously unknown mtDNA variants were less likely to be inherited than known variants, in which the level of heteroplasmy tended to increase on transmission. Variants in the ribosomal RNA genes were less likely to be transmitted, whereas variants in the noncoding displacement (D)–loop were more likely to be transmitted. MtDNA variants predicted to affect the protein sequence tended to have lower heteroplasmy levels than synonymous variants. In 12,975 individuals, we identified a correlation between the location of heteroplasmic sites and known D-loop polymorphisms, including the absence of variants in critical sites required for mtDNA transcription and replication.
We defined 206 unrelated individuals for which the nuclear and mitochondrial genomes were from different human populations. In these individuals, new population-specific heteroplasmies were more likely to match the nuclear genetic ancestry than the mitochondrial genome on which the mutations occurred. These findings were independently replicated in 654 additional unrelated individuals.
CONCLUSION
The characteristics of mtDNA in the human population are shaped by selective forces acting on heteroplasmy within the female germ line and are influenced by the nuclear genetic background. The signature of selection can be seen over one generation, ensuring consistency between these two independent genetic systems.